What is Amine Blush and Amine Bloom in Epoxy Art Resins?
Amine Blush and Amine Bloom are surface defects that negatively affect both the aesthetic and physical performance of epoxy art resin projects. And because epoxy art resin is used to create magnificent pieces of work, it's important we try to eliminate all factors which compromise the finished result! I hope this article explains this phenomenon in a way which is easy to understand but also educates the reader about the world of epoxy resin formulations!
Amine Blush is the most common type of surface defect experienced by Art Resin crafters, and something we regularly see being posted in forums, such as our Vista Art Resin Forum.
Simply put, Amine Blush is caused by a reaction of the Hardener part of the resin with moisture and carbon dioxide in the air.
More scientifically, Clive Hare explains “amines react with atmospheric carbon dioxide and moisture to form a carbamate that can produce a severe surface blush.”1 in the text Protective Coatings: Fundamentals of Chemistry and Composition 1998.
The occurence of Amine Blush can often be called crystallisation, because the bicarbonate salt which results from the reaction appears as crystals.
We try to prevent the occurrence of Amine Blush and Amine Bloom at the formulation stage of our Vista Art Resins.
Formulation (Science Bit):
Primarily, we do this by using adducted curatives, which are less prone to blush than alternative curatives because the primary amine hydrogens are pre-reacted with epoxy resin (and therefore not available to react with surface moisture and atmospheric carbon dioxide). Second, we use di-functional diluents (in place of more industry common monofunctional diluents) which have a high reactivity with amine hydrogens and so reduce free amines available to react with CO2 & H2O.
But even with an expertly formulated product such as Vista, blushing can still occur if the environment is susceptible for blushing to occur. 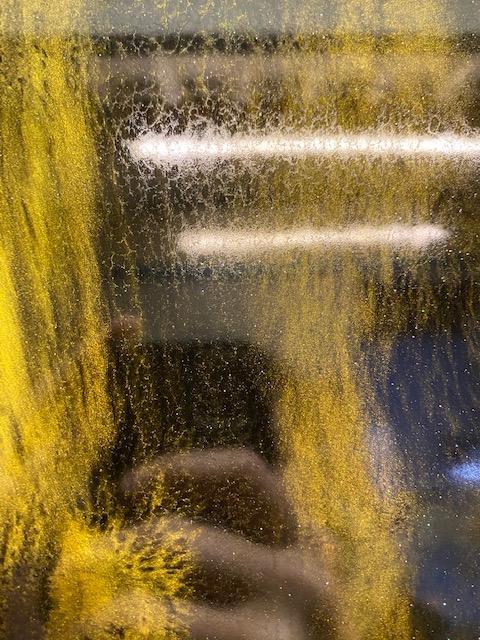
Photographic example of Amine Blush as experienced by some of Art Resin Crafters.
Conclusion:
Low temperature and high humidity combine to create condensation. Condensation enables the reaction of moisture and atmospheric carbon dioxide with the hardener component of the epoxy, creating Amine Blush. In addition, low temperatures elongate the curing process and extend the window available for the Amine Blush reaction to take place.
Amine Bloom:
Amine Blooming is slightly different to Amine Blush, although both can often occur together. Amine Bloom is caused by water-soluble compounds migrating to the surface drawn up by surface moisture. Blooming will appear on the surface as sticky deposits which typify Amine Blooming and separate this occurrence from Amine Blush.
Here is an example of Amine Bloom, typified by a sticky, oily surface.
In addition, the compounds that have migrated to the surface will contain an amount of Hardener component, and so if you’ve been reading this blog post carefully you should know what happens with the hardener component exposed to moisture and atmospheric carbon dioxide!
That is right, Amine Blush will occur! The hardener component will react with the surface moisture and carbon dioxide to create Amine Blush. Often, Amine Blush and Amine Blooming will occur together.
Here is an example of what appears to be Amine Bloom in the centre, and Amine Blush crystallisation further out. Both Amine Blush and Blooming occuring together.
You can read a blog explaining ways to prevent and fix Amine Blush and Blooming from our guest blogger and forum moderator Liz Hoyland - coming soon!
Hope you enjoyed the article , and feel a little more informed.
Sean Scott
Managing Director
3 Hare, C.H. Protective Coatings: Fundamentals of Chemistry and Composition, Society for Protective Coatings, Pittsburg, PA, 1998, p. 210.







